Exploiting Cobalt(II) Amide Complexes in Deprotonative Metalation of Fluoroaromatic Molecules
Fluoroaromatic compounds are increasingly heavily employed as scaffolds in agrochemicals and active pharmaceutical ingredients.[1] One of the most powerful methods for the incorporation of these molecules in more complex molecular scaffolds is deprotonative metalation. Typically these reactions have been the exclusive domain of group 1 and group 2 organometallics.[2] On the other hand, earth-abundant transition metals such as cobalt have shown excellent potential to selectively functionalize these molecules via C-H and C-F bond activation.[3]
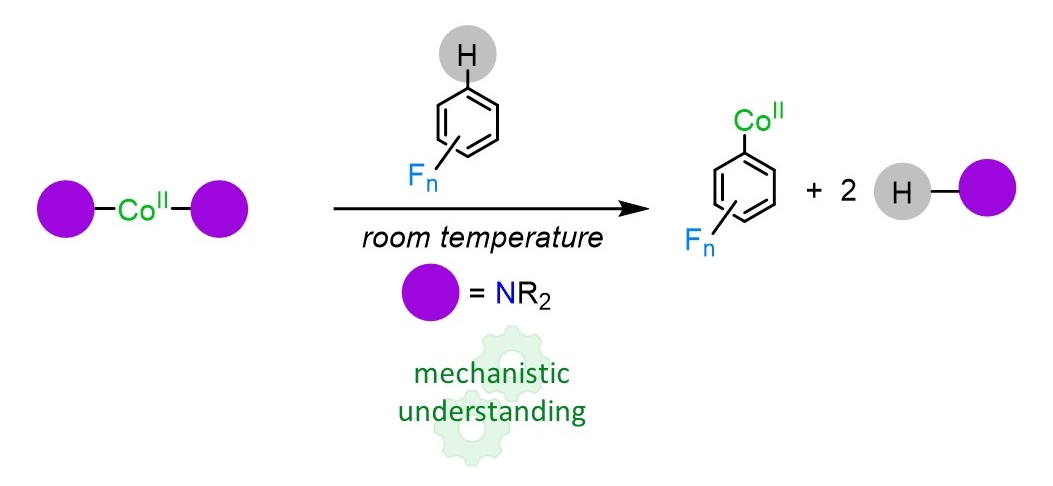
Breaking new ground in this field, in this contribution, we present the synthesis and the reactivity potential of a new Co(II) amide complex. While deprotonation of fluoroarenes using conventional lithium bases can be challenging due to the exceptionally fragility of generated intermediates,[4] the cobaltation of a range of different fluoroarenes proceeds regioselectively enabling the isolation of Co-H exchange products while operating at room temperature.
Combining the isolation of key metalated intermediates with theoretical calculations, this work exploits the potential of a new Co(II) base in a field widely dominated by main group element complexes, opening up for possible future development of new chemical transformations in this area of research.
[2] D. Robertson, M. Uzelac, R. E. Mulvey, Chem. Rev. 2019, 119, 8332
[3] a) J. V. Obligacion, M. J. Bezdek, P. J. Chirik, Am. Chem. Soc. 2017, 139, 2825; b) T. R. Dugan, X. Sun, E. V. Rybak-Akimova, O. Olatunji-Ojo, T. R. Cundari, P. L. Holland, J. Am. Chem. Soc. 2011, 133, 12418; c) O. Eisenstein, J. Milani, R. N. Perutz, Chem. Rev. 2017, 117,8710
[4] Schlosser, L. Guio, F. Leroux, J. Am. Chem. Soc. 2001, 123, 3822