Development of New Synthetic Approaches Towards Acridinium Salts and Their Applications
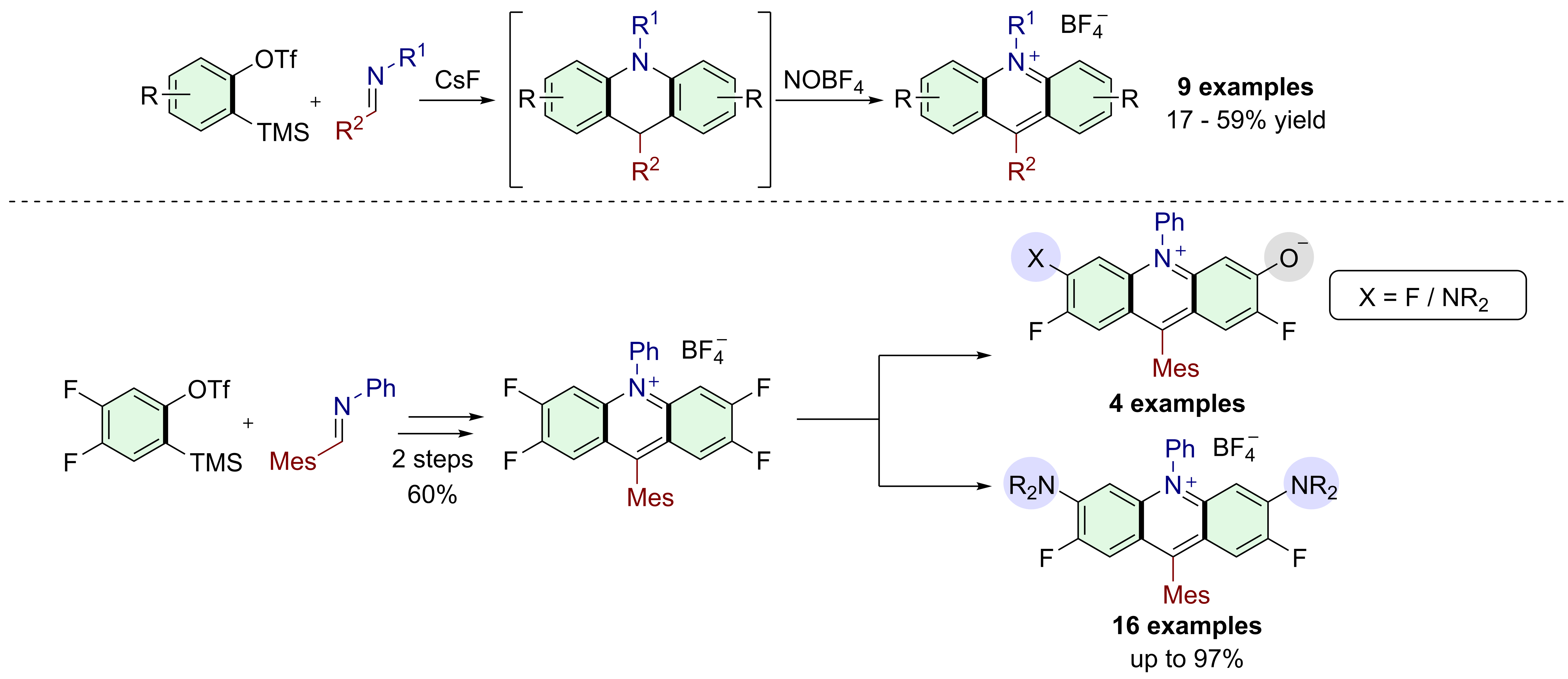
Photoredox catalysis has experienced significant advances over the last decade to become a valuable tool for otherwise intractable transformations under mild conditions.[1] In this context, short and modular synthesis of diverse photocatalysts with different photochemical properties is an important instrument for the further development of the field. As a sustainable replacement for the precious-metal photoredox catalysts, aсridinium salts have emerged as valuable organic cationic photocatalysts due to their favorable photophysical features.[2,3] We herein describe a new short two-step route to acridinium dyes comprising aryne-imine-aryne coupling combined with subsequent oxidation.[4, 5] This strategy was also applied for the preparation of a key tetrafluorinated acridinium salt which served as a linchpin intermediate for the late-stage diversification by nucleophilic aromatic substitution. Diverse acridinium derivatives were prepared with high yields using this methodology including aza-rhodols as a novel class of acridinium photocatalysts.[6] The obtained photocatalysts were successfully applied for organocatalytic C-N cross-coupling. Furthermore, the modularity of the developed synthetic strategy allowed the preparation of the acridinium derivatives with efficient intersystem crossing that were suitable for the polyarene reduction and triplet-triplet upconversion.[7]
[2] C. Fischer, C. Kerzig, B. Zilate, O. S. Wenger, C. Sparr, ACS Catal. 2020, 10, 210–215.
[3] V. Hutskalova, C. Sparr C., Science of Synthesis Knowledge Updates, (2022) 1, 1; 10.1055/sos-SD-115-00850.
[4] V. Hutskalova, C. Sparr, Synlett 2021, 32, DOI; 10.1055/s-0040-1720349.
[5] V. Hutskalova, C. Sparr, Helvetica 2021, e2100182.
[6] V. Hutskalova, C. Sparr, Org. Lett. 2021, 23, 5143–5147.
[7] B. Pfund‡, V. Hutskalova‡, C. Sparr, O. S. Wenger, unpublished results.
(‡ B. F. and V. H. contributed equally)