[99mTc(η6-pharm)2]+ synthesis with direct labelling of pharmaceuticals in water
Sandwich complexes with unchanged pharmaceuticals are unprecedented in bioorganometallic or radiopharmaceutical chemistry. We recently prepared a larger series of homo- and hetero sandwiches with Tc with direct labeling of arene in water.1
We will now present the direct labelling of pharmaceuticals with 99mTc through η6-coordination to phenyl rings and the confirmation of the structures with the rhenium homologues.2 Pharmaceuticals with an arene and simple organic functions were first chosen such as carbazole and indole (unit presents in some anticancer drugs)3, 4, 4-methoxyphenol (drug used in skin depigmentation), lidocaine (local anesthetic), but also more complexes drugs like lenalidomide (anticancer), erlotinib and gefitinib (tyrosine kinase inhibitor). (Figure).
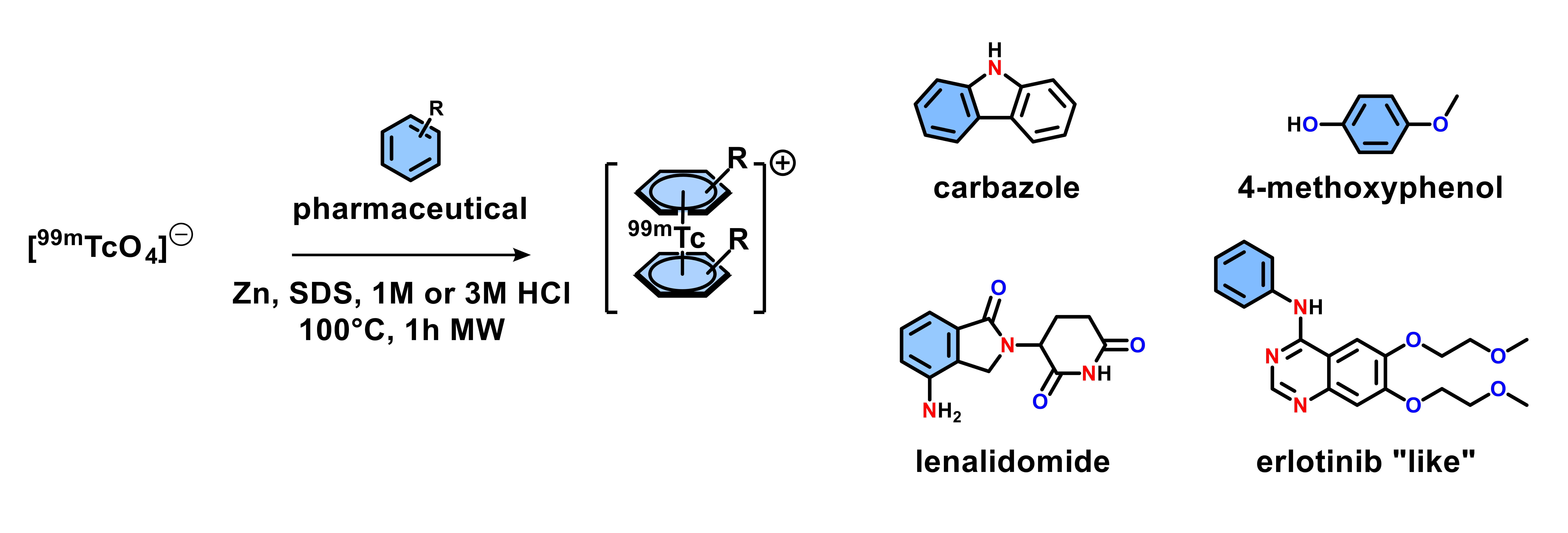
Figure. General scheme of the reaction towards [99mTc (η6-pharm)2]+.
Selected complexes display planar chirality since the two planes formed by the ligands are not equal; a feature that is very rare for rhenium or technetium if ever, but common for ferrocene or Ru-sandwich complexes.1,5-7.
The direct labelling of pharmaceuticals with 99mTc through η6-coordination to phenyl rings and the confirmation of the structures with the rhenium homologues opens a path into a wide variety of novel radiopharmaceuticals and molecular theranostics.
[1] Qaisar Nadeem, Giuseppe Meola, Henrik Braband, Robin Bolliger, Olivier Blacque, Daniel Hernandez Valdes, Roger Alberto, Angew. Chem. Int. Ed. 2020, 59, 1197–1200.
[2] Qaisar Nadeem, Federica Battistin, Olivier Blacque, Roger Alberto, Chem. Eur. J. 2022, 28, e20210356.
[3] Samar Issa, Anthony Prandina, Nicolas Bedel, Pål Rongved, Saïd Yous, Marc Le Borgne, Zouhair Bouazizb, J. Enzyme Inhib. Med. Chem. 2019, 34, 1321–1346.
[4] Jyothi Dhuguru, Rachid Skouta, Molecules 2020, 25(7), 1615.
[5] Ross A. Arthurs, Christopher C. Prior, David L. Hughes, Vasily S. Oganesyan, Christopher J. Richards, Organometallics 2018, 37, 4204–4212.
[6] Wei-Ping Deng, Xue-Long Hou, Li-Xin Dai, Yi-Hua Yua Wei Xia, Chem. Commun. 2000, 285.