Anticancer rhenium di- and tricarbonyl complexes and synthesis of new α-diimine rhenium dicarbonyl complexes
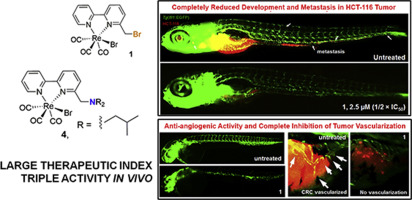
In our effort to discover novel selective and non-toxic agents effective against CRC, we synthesized a series of rhenium(I) tricarbonyl-based complexes with increased lipophilicity. Two of these novel compounds were discovered to possess remarkable anticancer, anti-angiogenic and antimetastatic activity in vivo (zebrafish-human HCT-116 xenograft model), being effective at very low doses (1-3 μM). At doses as high as 250 μM the complexes did not provoke toxicity issues encountered in clinical anticancer drugs (cardio-, hepato-, and myelotoxicity). In vivo assays showed that the two compounds exceed the anti-tumor and anti-angiogenic activity of clinical drugs cisplatin and sunitinib malate, and display a large therapeutic window.[1]
In another study, we reported a rhenium(II) dicarbonyl complex, which displayed better cytotoxicity against MCF-7 breast cancer cells than cisplatin.[2] We investigated later new synthetic routes to aerobically stable and substitutionally labile α-diimine rhenium(I) dicarbonyl complexes. The molecules were prepared in high yield from the cis–cis–trans-[Re(CO)2(tBu2bpy)Br2]− anion ( where tBu2bpy is 4,4′-di-tert-butyl-2,2′-bipyridine), which could be isolated from the one electron reduction of the corresponding 17-electron complex. Ligand substitution of Re(I) complexes proceeded via pentacoordinate intermediates capable of Berry pseudorotation. In addition to the cis–cis–trans-complexes, cis–cis–cis- (all cis) isomers were also formed. [Re(CO)2(tBu2bpy)Br(L)] complexes may be considered as synthons for the preparation of a variety of new stable diamagnetic dicarbonyl rhenium cis-[Re(CO)2]+ complexes, offering a convenient entry in the chemistry of the core.[3]
[2] Jeremie Rossier, Daniel Hauser, Emmanuel Kottelat, Barbara Rothen-Rutishauser, Fabio Zobi, Dalton Trans., 2017, 7, 2159.
[3] Kevin Schindler, Aurélien Crochet, Fabio Zobi, RSC Adv., 2021, 13, 7511.