Single-molecule profiling of PRC1 ubiquitination dynamics in defined chromatin states
Each cell of a eukaryotic organism carries universal genetic information despite its functional diversity. The DNA is organized as chromatin fiber, i.e. long arrays of nucleosomes. Cell-type-specific gene expression patterns have to be established to develop a diverse set of cell types in multicellular organisms.
Dynamic regulation of transcription is orchestrated by a large cohort of enzymes, among which chromatin modifiers or ’writers’ install histone post-translational modifications (PTMs) controlling the recruitment of chromatin ’readers’. A specific subset of chromatin modifiers includes Polycomb group (PcG) proteins, which drive the inheritance of a repressed chromatin state during development and cell differentiation while preventing abnormal oncogenic transformations.
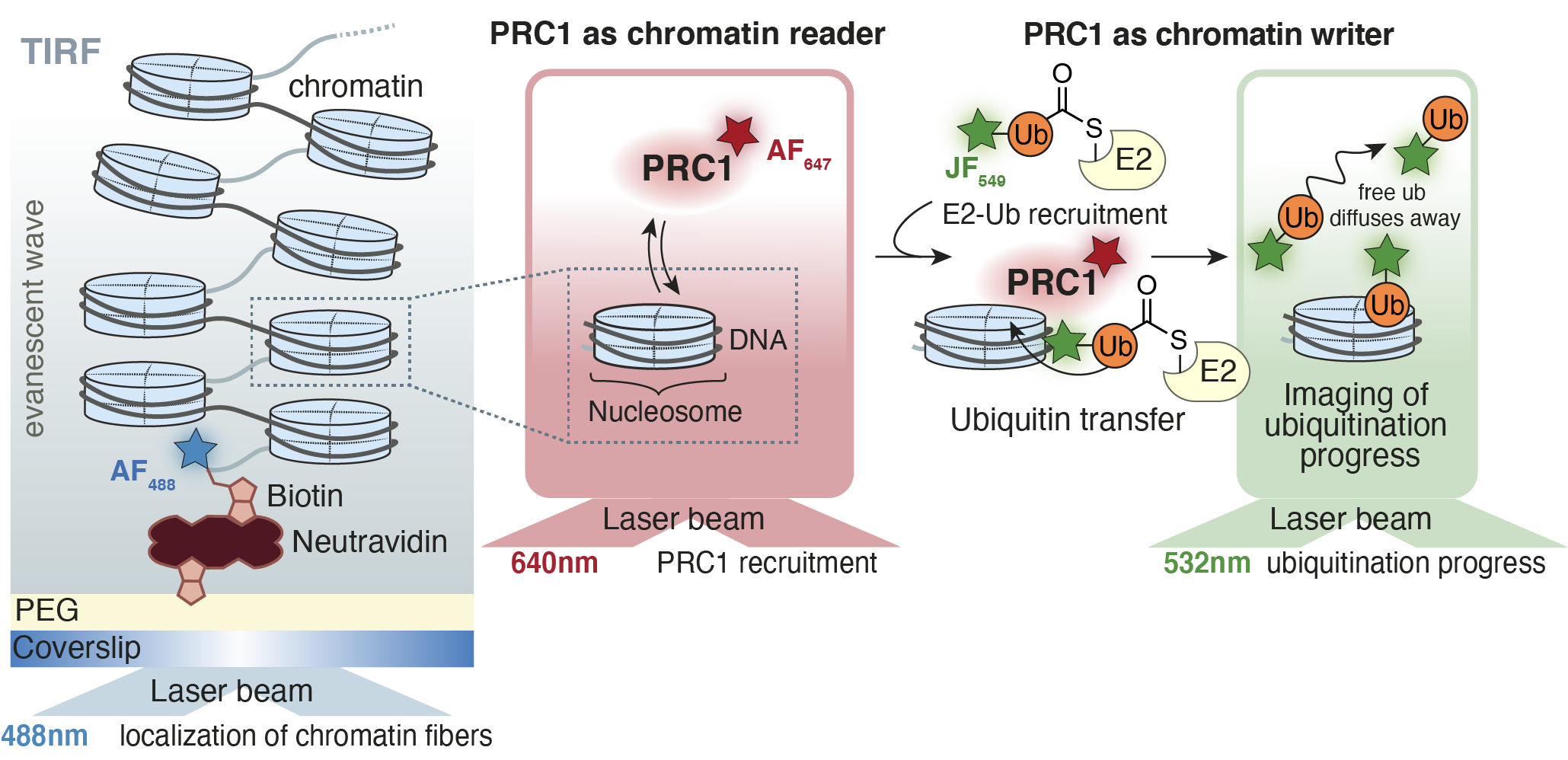
PcG members Polycomb Repressive Complex (PRC) 1 function as H2A-specific E3 ligases that establish gene repression. Variant PRC1 is involved in most of the ubiquitin deposition in the context of repressed chromatin, without the requirement of primary installation of additional PTMs. We hypothesize that chromatin modification, i.e. ubiquitination, by PRC1 is controlled by its recruitment dynamics on underlying chromatin and its subunit composition.
Here, we present a single-molecule method, which allows us to directly observe PRC1 binding dynamics on immobilized chromatin fibers in real-time. Consequently, we introduce ubiquitin-conjugating enzyme E2 which is loaded with fluorescently labeled ubiquitin. The preloaded E2-Ub is recognized by PRC1 driving H2AK119 ubiquitination. This allows us to gain a mechanistic view of ‘reading’ and ‘writing’ by PRC1 in real-time and elucidate its contribution to gene regulation.