Synthesis of short peptides in 384-well plates for generating large libraries of macrocyclic compounds
Macrocycles have raised much interest in the pharmaceutical industry due to their ability to bind challenging targets. However, the development of macrocyclic ligands to new disease targets is hindered by the limited availability of large, structurally diverse macrocyclic compound libraries for high-throughput screening.
In order to generate large libraries of macrocycles, our laboratory has recently introduced a new strategy in which "n" short linear peptides are combinatorially cyclized with "m" different chemical linkers to generate "m x n" different macrocyclic compounds.[1, 2] A bottleneck in applying the established strategy is the synthesis of large numbers of short peptides. The peptide synthesizer that we use – being the one having the highest throughput on the market - can produce 4 x 96 in four microtiter plates, which limits the synthesis to 384 peptides per run. The synthesizer holds reagent containers for a maximum of 31 different amino acids, which also limits the chemical and structural diversity of peptides that can be produced.
Herein, we have developed hard- and software to upgrade the microtiter plate based solid-phase peptide synthesizer. Now, the peptides can be synthesized in four 384-well plates, allowing the synthesis of 1,536 peptides in one run. The number of possible buildings blocks was increased from 31 to 395 derivatives. Moreover, we have developed practical tools for the rapid and reliable resin loading to 384-well plates. The short peptides synthesized in the 4 x 384-well plates were of high purity.
With the new peptide synthesis capacity, it has become possible to synthesize large combinatorial libraries comprising ten-thousands of macrocyclic compounds. The peptide synthesis in 384-well plates may be also attractive for other applications beyond macrocycle drug development, such as for antibody epitope scanning, epitope mimetic development, peptide ligand development, and peptide-substrate screening.
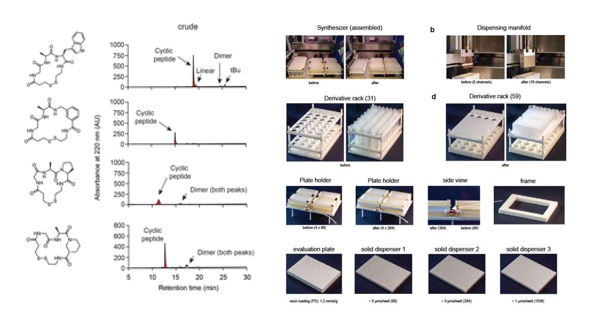
[2] Sevan Habeshian, Ganesh A. Sable, Mischa Schüttel et al., ACS Chem. Biol. 2022, 17, 181−186.