Chemodivergent Asymmetric Synthesis via Catalytically Formed Chiral Auxiliary
Asymmetric catalysis remains as one of the most efficient ways to obtain enantioenriched products. This approach has culminated in Nobel prizes for asymmetric transition metal catalysis and organocatalysis in 2001 and 2021 respectively. However, catalytic asymmetric reactions are sensitive to the substrate structure and often require specialized reagents or additives [1]. In addition, for each chemically distinct transformation a different fine-tuned catalyst is required, because the catalyst is responsible for both reactivity and stereoselectivity.
We speculated that the reactivity and stereoselectivity could be decoupled by first introducing a chiral auxiliary using asymmetric catalysis, and then performing chemodivergent, robust transformations in a diastereoselective manner. Here we report a Pd-catalyzed enantioselective tethered carboetherification of propargylic amines using a trifluoroacetaldehyde derived molecular tether and aryl iodides as electrophiles (Scheme 1) [2]. The catalytically formed auxiliary was used to control facial selectivity in multiple chemically distinct transformations of the C-C double bond: hydrogenation, cyclopropanation and epoxidation [3]. In all cases challenging asymmetric reaction on tetra-substituted olefin was achieved using very simple conditions – heterogenous hydrogenation, cyclopropanation using free carbene and epoxidation with mCPBA. The obtained products serve as precursors to medicinally relevant amino alcohols or alpha amino ketones.
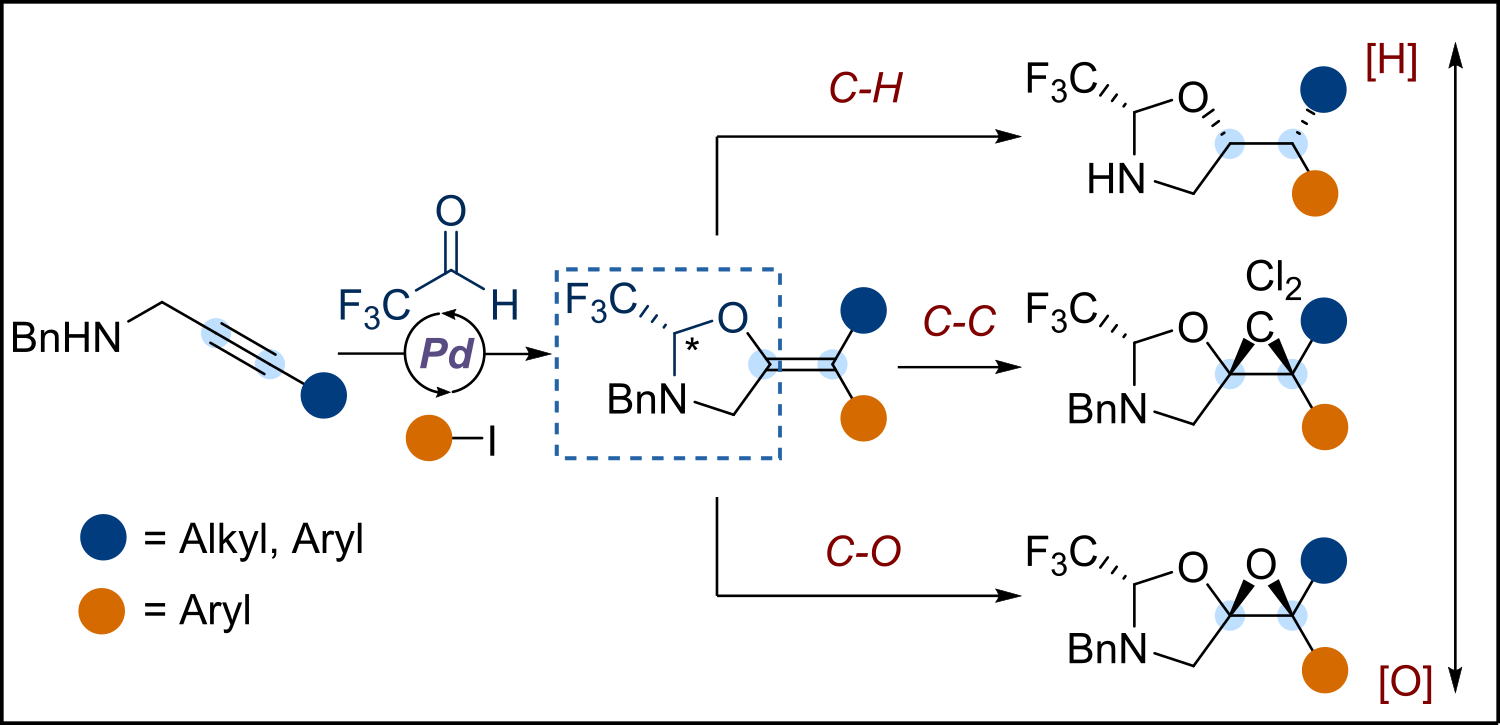
Scheme 1. Chemodivergent asymmetric synthesis via catalytically formed chiral auxiliary.
[2] Luca Buzzetti, Mikus Purins, Phillip D. G. Greenwood, Jerome Waser, J. Am. Chem. Soc., 2020, 142, 17334-17339.
[3] Mikus Purins, Jerome Waser, Angew. Chem. Int. Ed., 2022, 61, e202113925.